- Community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA) abscesses are best managed surgically; postprocedure antibiotics do not substantially improve outcomes. Cure rates with incision and drainage alone are at least 90% (A).
- If incision and drainage fail to promote healing within 7 days, the oral antibiotics of choice are trimethoprim-sulfamethoxazole and tetracycline (C).
- Eradication of nasal carriage of CA-MRSA is generally not useful in preventing spread of clinical MRSA infections in communities (B).
Strength of recommendation (SOR)
- Good quality patient-oriented evidence
- Inconsistent or limited-quality patient-oriented evidence
- Consensus, usual practice, opinion, disease-oriented evidence, case series
A previously healthy law student arrives at your office complaining of “abdominal pain.” You discover on examination that she has an erythematous, indurated, and tender 3-cm lesion on her suprapubic region. The lesion has no point, but its center is boggy. The patient’s temperature is normal. Would you give her an antibiotic? Would you cover immediately for community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA)? What other factors might influence your decision?
The incidence of MRSA is increasing in communities across the United States, challenging our assumptions about evaluation and management of skin and soft-tissue infections. In this article, I outline a rational approach to managing patients who have lesions likely to have been caused by CA-MRSA (TABLE).
TABLE
Suspect CA-MRSA? Consider this treatment approach6,7
| CLASS | PATIENT CRITERIA | MANAGEMENT | ANTIBIOTIC CHOICES |
|---|---|---|---|
| 1 | Afebrile and healthy; lesion nonfluctuant | If no drainable abscess, give common first-line antibiotic for SSTI; reassess for response | Semisynthetic penicillin, oral first- or second-generation cephalosporin, macrolide, clindamycin |
| 2 | Fluctuant or pustular lesion <5 cm; with or without fever | Surgical drainage of abscess if possible. Use I&D presumptively for MRSA and monitor closely for response; inpatient management may be indicated | Trimethoprim-sulfamethoxazole, tetracycline, clindamycin |
| 3 | Toxic appearance or at least 1 unstable comorbidity or a limb-threatening infection; lesion >5 cm | Hospital admission with broadspectrum antibiotics for MRSA coverage; consider infectious disease consultation | Broad-spectrum, including vancomycin |
| 4 | Sepsis syndrome or life-threatening infection (necrotizing fasciitis) | Above plus aggressive surgical debridement | Above with infectious disease guidance |
| CA-MRSA, community-acquired methicillin-resistant Staphylococcus aureus; I&D, incision and drainage; MRSA, methicillin-resistant Staphylococcus aureus; SSTI, skin and soft-tissue infection. | |||
When to suspect MRSA skin infection
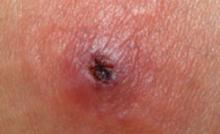
Patients with CA-MRSA skin infection often report a “spider bite,” as lesions appear suddenly and unexpectedly in areas without a history of trauma.1 The lesions very often are pustular with central necrosis, and there may be purulent drainage, redness, tenderness, and palpable fluctuance. CA-MRSA can cause impetigo, but the often benign nature of this clinical infection makes management decisions less crucial. CA-MRSA skin lesions can occur anywhere on the body, though most often they appear in the axillae or the groin and buttocks. Patients may or may not have a fever.
Individuals who are at increased risk for CA-MRSA disease include users of health clubs or participants in contact sports, men who have sex with men, children younger than 2 years of age, users of intravenous drugs, military personnel, and prisoners.2,3 However, the absence of these factors in a patient with a skin or soft-tissue infection does not rule out MRSA.4
Regardless of the lesion’s appearance or the patient’s epidemiologic history, consider CA-MRSA if its prevalence in your community reaches 10% to 15%.
- Community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA) causes up to 74% of purulent skin and soft-tissue infections in communities throughout the United States.1 By definition, this infection occurs in patients who have not been hospitalized and have not undergone medical procedures within the prior year.12
- The annual incidence of CA-MRSA was reported to be 18.0-25.7 cases per 100,000 population between 2001 and 2002.13 Clusters of CA-MRSA have been identified among Alaskan natives, Native American Indians, and Pacific Islanders.12
- Most often this organism causes skin and soft-tissue infections, though cardiac, respiratory, blood, and bone infections can also occur.14
- CA-MRSA species are genotypically distinct from hospital-acquired MRSA. One marker for CA-MRSA, Panton-Valentine leucocidin (PVL), is most often detected in cases of severe and systemic infection, and it may be a virulence factor.15 However, the presence of PVL does not necessarily correlate directly with antibiotic resistance.
- Historically, CA-MRSA was primarily resistant to beta-lactams and erythromycin. More recent strains have also demonstrated resistance to tetracycline and clindamycin.
- Retrospective analyses show that patients with CA-MRSA tend to receive inadequate initial antibiotic coverage, and, independent of this, they tend to have worse clinical outcomes than those infected by methicillin-sensitive strains.16
Hospitalize any patient who exhibits fever or hypothermia, tachycardia greater than 100 beats per minute, or hypotension with a systolic blood pressure <90 mm Hg or 20 mm Hg lower than baseline. A skin lesion >5 cm is also likely to require hospitalization and parenteral antibiotics.5
Treatment: Incision and drainage most important